Have you ever skipped medication because you couldn’t swallow a tablet? Or struggled to give medicine to a child who refuses to take it?
A new dosage form—oral dissolving film (ODF)—is transforming this experience. Ultra-thin and fast-dissolving, it melts on the tongue within seconds without water and delivers rapid therapeutic effects. This is not marketing hype; it is grounded in pharmacokinetics, mucosal absorption science, and modern pharmaceutical engineering.
In this article, we break down how oral dissolving films are absorbed, why they act faster than conventional tablets, and what makes them a scientifically validated drug delivery system.
1. The Natural Limitations of Traditional Oral Tablets
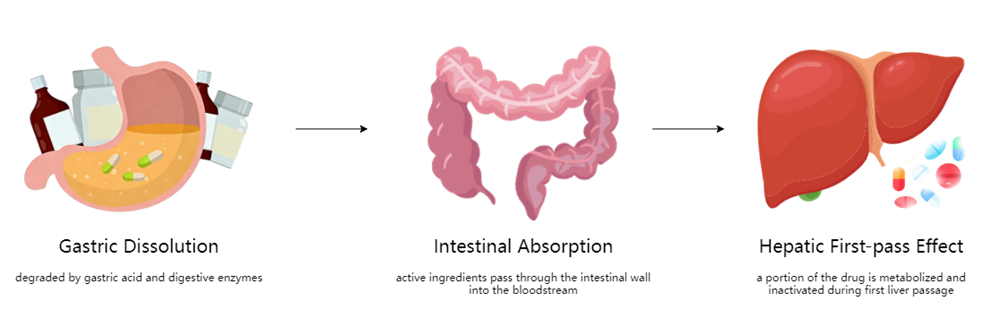
Conventional tablets and capsules must pass through several physiological steps before becoming effective:
hysiological Steps Tablets And Capsules Before Becoming Effective
- Disintegration in the stomach – broken down by gastric acid and digestive enzymes
- Intestinal absorption – active ingredients cross the intestinal wall into systemic circulation
- First-pass metabolism in the liver – a portion of the drug is metabolized and inactivated
This entire process typically takes 30 minutes to 2 hours to reach effective plasma concentrations. More importantly, the first-pass effect in the liver can significantly reduce bioavailability. For certain drugs, less than 50% of the administered dose ultimately reaches systemic circulation in active form.
This pathway creates both a time delay and efficiency loss.
2. The “Shortcut” of Oral Dissolving Films: Direct Mucosal Absorption
The core advantage of oral dissolving films lies in their ability to bypass the gastrointestinal tract and liver, utilizing a more direct absorption route—the oral mucosa.
Why Is the Oral Cavity an Effective Absorption Site?
The sublingual and buccal mucosa offer three major advantages:
1.Rich vascularization – dense capillary networks enable rapid entry into systemic circulation
2.No keratinized barrier – unlike skin, the mucosa allows easier molecular permeation
3.Near-neutral pH environment – reduces chemical degradation risk
When placed on the tongue, the film rapidly dissolves in saliva. The drug diffuses across the mucosal epithelium and enters the bloodstream within minutes.
u The Critical Breakthrough: Avoiding First-Pass Metabolism
This is the fundamental reason ODFs act faster and often more efficiently than tablets.
Drugs absorbed via traditional oral routes first pass through the liver, where metabolic enzymes reduce the active fraction. In contrast, drugs delivered via oral dissolving film are absorbed through oral veins and drain directly into the internal jugular vein → superior vena cava → heart → systemic circulation.
The liver is bypassed during initial absorption.
Clinical studies have shown that for certain drugs—such as:
·Ondansetron
·Montelukast sodium
bioavailability may increase by 2–3 times when delivered via oral dissolving film compared to conventional tablets.
This means that at the same dose, the body receives more active drug—resulting in faster onset and potentially improved therapeutic efficiency.
3. Data-Driven Performance: Faster Tmax, Better Compliance
Oral dissolving film is not a conceptual innovation—it is a clinically validated drug delivery platform.
The U.S. Food and Drug Administration has approved dozens of ODF-based medications across multiple therapeutic areas, including psychiatry, pediatrics, and oncology supportive care.
Pharmacokinetic studies consistently demonstrate:
Shorter Tmax (time to peak concentration) compared to tablets
Oral Dissolving Film
Example: an antiemetic ODF shows Tmax ≈ 15 minutes vs. 60 minutes for tablet form
·Improved patient compliance, particularly in pediatric and geriatric populations
·No swallowing required
·Pleasant taste options
·No choking risk
Studies report compliance improvements exceeding 70% in certain populations.
The rapid onset of oral dissolving film is therefore not perceived—it is scientifically measurable and reproducible.
4.From Science to Scalable Manufacturing
Advanced formulation alone is not enough. The commercial success of oral dissolving films depends heavily on precision manufacturing.
ODFs demand strict control over:
·Film thickness uniformity
·Drug loading accuracy
·Moisture barrier protection
·Mechanical integrity
Even minor deviations can affect dissolution rate and therapeutic performance.
4.1Film Casting Precision
Aligned Machinery’s oral dissolving film casting systems utilize high-precision coating technology combined with stable multi-zone drying control. Thickness deviation can be controlled within ±2 μm, ensuring uniform drug distribution and consistent film structure.
OZM - Oral Strips Film Making Machine
The system supports multiple formulation types and provides stable output for downstream slitting and packaging, meeting both R&D and large-scale production requirements.
4.2High-Barrier Packaging Protection
ODFs are highly sensitive to humidity and oxygen. Aligned Machinery’s fully automatic ODF packaging systems integrate cutting, forming, and sealing in a single line, with speeds up to 180 films per minute.
KFM - Oral Strips Packing Machine
Aluminum-plastic composite barrier structures effectively protect against moisture ingress and oxidative degradation, extending shelf life and ensuring long-term product stability. The entire line operates under pharmaceutical-grade GMP standards, enabling sustainable, high-quality mass production.
Conclusion: When Science Makes Medicine Simpler
The rise of oral dissolving film is not accidental. It reflects a deep understanding of human physiology and pharmacokinetics. A thin film solves swallowing challenges, enhances drug efficiency, and embodies patient-centered healthcare innovation.
Behind every transparent strip lies the integration of pharmaceutical science, materials engineering, and precision manufacturing.Science does not complicate healthcare—it makes it simpler, faster, and more effective.
Aligned Machinery is a leading manufacturer of pharmaceutical equipment with over 20 years of experience. Our machines are used globally by pharmaceutical and nutraceutical companies. Known for quality, reliability, and international compliance, we help partners enhance efficiency and achieve sustainable growth.
For more information or project discussions, contact us
+86 13967712128
*Note: The mechanisms described apply to approved oral dissolving film pharmaceuticals and compliant functional health products. Always follow medical advice or product instructions for specific use.



Oral thin film making machine is typically designed for manufacturing orally disintegrating films, fast dissolving oral films and breath freshening strips. It is particularly suitable for oral hygiene and food industries.
These equipment adopts frequency conversion speed control and automatic control technology of machine, electric, light and gas, and innovates the design according to the “GMP” standard and the “UL” Safety Standard of the pharmaceutical industry.
Aligned KFM-300H High Speed Oral Disintegrating Film Packaging Machine is designed for cutting, integrating, compounding, and sealing film-like materials, catering to pharmaceutical, healthcare, food, and other industries.
Oral dissolving Film Packaging Machine features variable frequency speed regulation technology and an automatic control system that integrates machinery, electricity, light, and gas for precise adjustments according to production requirements. This ensures improved stability, reliability, and smooth operation, while simplifying equipment operation and reducing production debugging complexity.
Oral thin film making machine is typically designed for manufacturing orally disintegrating films, fast dissolving oral films and breath freshening strips. It is particularly suitable for oral hygiene and food industries.
These equipment adopts frequency conversion speed control and automatic control technology of machine, electric, light and gas, and innovates the design according to the “GMP” standard and the “UL” Safety Standard of the pharmaceutical industry.
Post time: Jan-05-2026